|
It quantitatively explains the emission and absorption line spectrums of hydrogen and hydrogen-like atoms.Įach spectral line in absorption or emission spectrum can be associated to transition of states by the electron of a particular hydrogen atom.īohr's model also shows that the intensity of each spectral line depends upon the frequency or wavelength of photons absorbed or emitted. The energies of the stationary states of hydrogen-like ions are given by the expressionĪccording to Bohr's model, electrons are held in orbits by virtue of an electrostatic force, so that doesn't fall into the nucleus. Where n is the principal quantum number of the orbit and R H is Rydberg's constantīohr's theory also applies to other hydrogen-like ions, such as He ⁺, Li ⁺² and Be. The energy of a stationary state or orbit is given by the equation An electron moving in a circular orbit has an angular momentum equal to the product of its mass (m e ), linear velocity (v) and radius of orbit (r), which can be expressed as The Bohr model of the electronic energy levels of the hydrogen atom is based on four postulates: The electron in the atom has a fixed number of stationary. The frequency of the radiation emitted or absorbed can be represented by the equation,Ĥ. The energy gap between the two orbits is given by equationģ. Bohr found that an electron located away from the nucleus has more energy, and the electron which. Bohr’s model consists of a small nucleus (positively charged) surrounded by negative electrons moving around the nucleus in orbits. The K-shell of the Bohr diagram of Hydrogen has only 1 electron. This nucleus is surrounded by only one electron shell named K-shell. In going to a higher level (1) PE decreases, TE increases (2). Le modèle de Bohr de lhydrogène repose sur lhypothèse non-classique que les électrons tournent autour du noyau selon des couches ou orbites spécifiques. Rutherford explained the nucleus of an atom and Bohr modified that model into electrons and their energy levels. The Bohr Model of Hydrogen (H) has a nucleus with 0 neutrons and 1 proton. So each emission or absorption of radiation energy represents the electron transition from one stationery orbit to another. In Bohrs model of hydrogen atom, let PE represents potential energy and TE the total energy. Here’s how you can draw the bohr model of hydrogen step by step. The energy of an electron in the orbit remains constant until the electron absorbs energy to jump to a higher orbit or releases energy to move to a lower orbit. Electrons are held in orbits by an electrostatic force.Ģ. The electron in the hydrogen atom can move around the nucleus in circular paths of fixed radius and energy called orbits also called stationary states. four assumptions absorption & emission of photons hydrogen. It was based on the four postulates - Motion of electrons in an orbit, Fixed Energy of electrons, Transition of orbits by electrons, and Angular momentum of electrons.ġ. Bohr energy levels and transitions with labels Bohr model of the atom. 4) It was not explained the formation of chemical bonds.In 1913, Neils Bohr proposed an atomic model that quantitatively explained the structure of hydrogen atom and its spectrum. 3) Bohr theory was not explained the quantisation of angular momentum of an electron. 2) This model failed to account for the atomic spectra of atoms of more than one electron. The great success of the Bohr Model had been in explaining the spectra of hydrogen-like (single electron around a positive nucleus) atoms. `therefore L = m v r = (nh)/(2pi)` m =mass of electron, v= velocity of electron, r = radius of circular path, h = plank constant Limitations : 1) Bohr's model failed to account for splitting of line spectra (Zeeman effect). The Bohr model of the atom was replaced by the Quantum Mechanics Model based upon the Schrodinger equation in the 1920s. He +, Li 2+, Be 3+, which have only one electron. The model is also applicable to ions similar to hydrogen, e.g. Hydrogen atom is the simplest atom with one proton and one electron. d) the angular momentum of electron is multiple integral of `(L)/(2pi)`. Bohrs model holds good for the hydrogen atom. 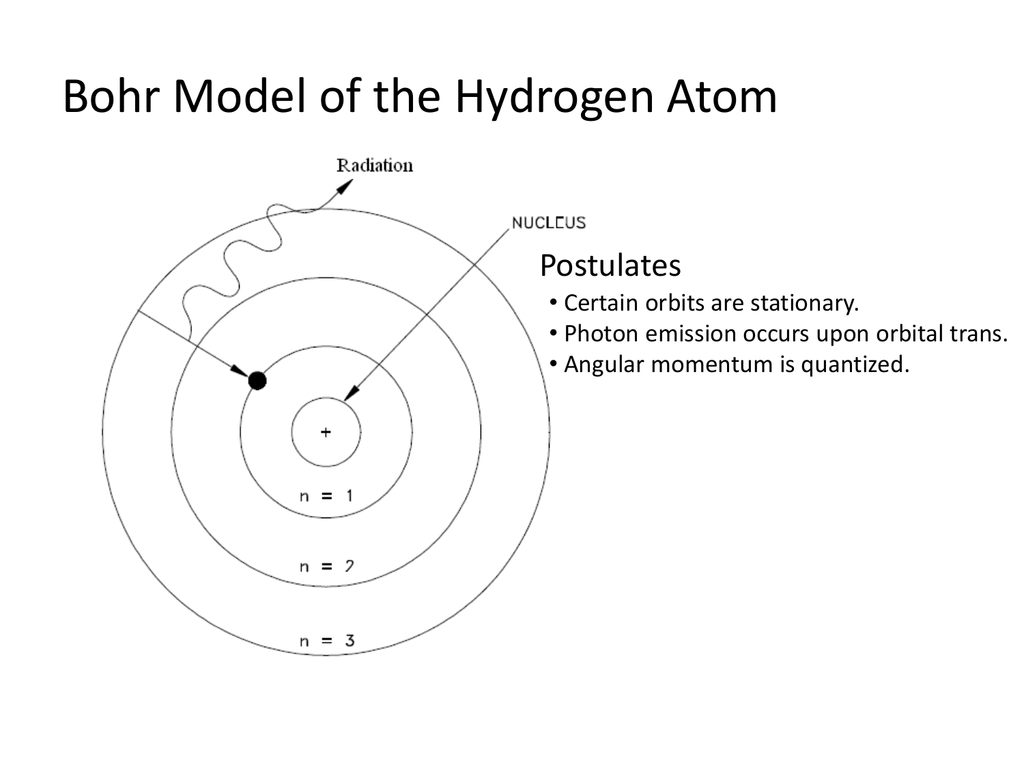
The states corresponding to these energies are called stationary states and the possible values of the energy are called energy levels. c) the energies of an electron in an atom can have only certain values `E_(1), E_(2), E_(3)`. b) when an electron jumps from a lower energy state to higher energy state, it absorbs energy or emits energy when such a jump occurs from a higher energy state to lower energy state. 
) at different distances from the nucleus. Solution : Niels Bohr proposed that a) electrons in an atom occupy stationary orbits of fixed energy (K, L, M, N.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
